Last week the headlines read “elusive bird hormone found” and “discovery of the elusive leptin in birds.” It has been great to see so much interest in research I have been involved with for several years, but our paper (Reference 1) and the press blurbs associated with it only tell a small part of the story.
To fill out the rest of the story, I am taking my friends, family and extended blog family on a behind-the-scenes tour of this research project from inception to publication. In this two-part series I will provide a primer on leptin biology, highlight the twists and turns of an ongoing research project and provide a personal perspective on our successful search for the “elusive leptin in birds.”

What is Leptin?
Where better to begin than with the star player of this drama – Leptin. Leptin is a hormone produced in adipose (fat) cells of animals that signals the brain to regulate appetite, metabolic rate and fat storage. An overly simple but useful model for understanding one way leptin can function goes something like this: as you build up fat you increase your leptin production which increases the amount of leptin circulating in your blood which in turns is read by your brain as a notice that you have enough fat reserves. The result is that hopefully your appetite for food is modified. So the general rule is; high leptin reduces appetite and low leptin stimulates appetite. Simple, right! You are probably asking yourself “where can I get my daily dose of leptin!” Unfortunately, although leptin really can help in some cases it doesn’t work quite that easily.
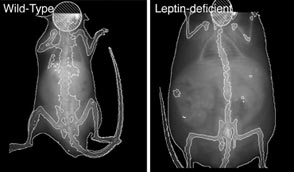
Leptin isn’t just a simple fat metabolism switch. This why leptin isn’t the magic weight loss pill though that it is marketed to be (LeptAway, LepinX, LeptiFit, LeptiLean etc..). We now know that leptin in addition to regulating appetite and fat metabolism also regulates reproduction, body temperature, growth, bone remodeling, immune function, and many other traits (reviewed in reference 2).
Because leptin plays a significant role in so many aspects of our physiology there has been considerable interest in understanding exactly how it works. Literally thousands of studies have been performed to study its effects on humans and many other vertebrates. Today there are leptin treatments for several human conditions and leptin production has been manipulated in many animals to induce either leaner or fatter animals for food production.
Most of what we know about leptin comes from our study of humans and mice. Most mammals seem to use leptin in similar ways though some extreme mammals like bats, whales and seals have likely modified their leptins for additional purposes. A growing number of studies of leptin outside mammalian systems tell us that the role of leptin may be very different in other animals. Therefore simply applying what we know about leptin from a mouse model to something like fishery production may not provide the expected outcome. A better understanding of the full range of lepton’s functions and capabilities will only come through a comparative approach which requires understanding the full range of leptin’s capabilities across a wide diversity of animals.
Who has leptin?
The leptin hormone is encoded by an approximately 500 base pair piece of DNA in our genome (on the long arm of chromosome #7). It was first characterized in mouse in 1994 and then in human. For many years it was not known if this hormone existed in non-mammals but eventually it was identified in a fish and a lizard. After finally identifying those DNA sequences it became apparent why it took 10 years to find them: the sequences of fish, lizards and mammals are far less than 50% similar to one another and therefore very difficult to identify as being the same gene.
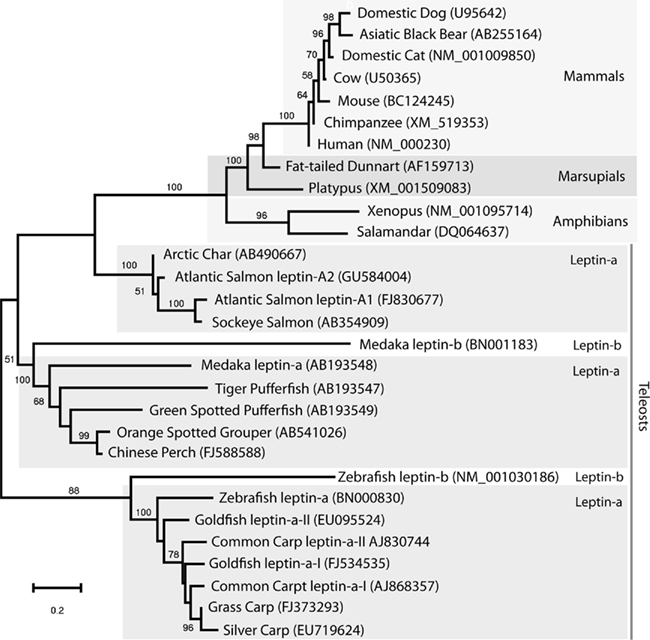
In the past decade numerous additional leptin sequences have been identified in fish and a few amphibians (see figure below). But there has been one glaring omission in the leptin universe: birds! Despite great interest by the poultry industry the decade long search for bird leptin and chicken leptin in particular, has yielded no definitive leptin hormone, hence, the “elusive leptin” from the title of our recent paper.
The elusive bird leptin?
Here is where the story starts to get a bit strange. There has been every reason to believe that birds produce leptin, not least of which is that if you inject human or mouse leptin into a chicken it has dramatic physiological effects including changing their appetite and body fat percentage. Why do they respond to leptin if we can’t find evidence they make leptin? They respond because some chicken cells express the leptin receptor proteins on their surfaces!
This chicken leptin receptor is very similar to human and other vertebrate leptin receptors which is why human leptin can interact with it. The leptin receptor is encoded by its own gene (DNA sequence) and that gene was discovered many years ago. Some of the basic units (amino acids) that construct a receptor protein are particularly important for maintaining the function of that protein. These amino acids are situated at sites that form the binding site with the leptin hormone thus creating the hormone-receptor connection (see figure below) that should trigger a response in the cell that has the receptor on its surface.

So chickens, and birds in general, have a gene that encodes for a receptor for the leptin hormone. This should strike you as peculiar. If chickens have receptors for the hormone surely they must be making the hormone, right? A reasonable question but let me provide a bit more background before exploring possible answers.
A year ago our group at the University of Akron published a paper that modeled leptin hormone and the leptin receptor binding site (eg. where the hormone and the receptor connect) using known sequences of both genes from many distantly related vertebrates. (Reference 4). That work showed that there are many specific amino acids that are conserved in the receptor and leptin hormone across all vertebrates. By conserved I mean that these are positions in these proteins that have not changed during the evolution of all vertebrates. Presumably they have not changed because these positions are so important for maintaining the function of the leptin hormone system that any mutation to these sites would have been quickly eliminated by natural selection. In other words, mutations may happen randomly within a gene but those that are harmful are non-randomly eliminated. The result is that organisms can come to have very different sequences over time for some portions of their genes while maintaining high degrees of similarity in other portion of the same gene.
So if leptin is important and the leptin receptor is so conserved that it will react to a human leptin hormone doesn’t that suggest that the birds must be making a leptin molecule to interact with the receptor? Why then has bird leptin hormone been so difficult to find?
There are several possibilities:
1) Leptin exists in birds but most of its sequence is so divergent from other animals that we just can’t recognize it when we see it. Fish were less than 20% similar in their primary sequence similarity with mammals so maybe birds also have radically different sequences.
2) Bird leptin as we know it doesn’t exist because the gene was lost in the first birds. At the same time another gene with similar characteristics, most importantly the same critical binding sites amino acids, evolved to take its place. This would not be unprecedented. There are known receptors for which two separate proteins can bind in which case if one of the proteins is mutated and eventually lost from the genome the other protein will take over the duties of providing the function of the lost gene. This is how one gene can be replaced by another gene over time.
3) Birds really don’t have leptin and their leptin receptors are simply a vestigial protein: they are made but don’t have a function. However, that receptor still works if we put leptin in a birds system. Alternatively, maybe some birds still use leptin and it’s only the birds we have looked at so far (we tend to be chicken-centric in our thinking) that have lost their leptin gene.
Finding a gene in a haystack
So, how might one find a gene of 500 base pairs in a genome of 2 to 5 billion total base pairs? There are two primary methods, 1) using the DNA sequence of a known gene like human leptin and using it to go fishing in another genome looking for a similar sequence, and 2) find the genes that are usually right next to your gene of interest and look to see if your gene is there just like it is in other genomes.
The first method could be confounded by target sequences that are so different that the gene is unrecognizable at the DNA sequence level. This is why fish and reptile leptins were not found for many years. The first leptin sequence we had was from mouse and we used that sequence then to hunt for other leptins in other animals. Because all mammals are thought to be related to one another via a relatively recent common ancestor it is not surprising that all their leptins sequences are quite similar. But fish and reptiles are much further removed from mammals and their DNA sequences for leptin turned out to be so different that having a mammal leptin as a starting point was of little use for finding a fish leptin sequence. So researches turned to the second technique called gene synteny.
The idea of using gene order comes from the evolutionary assumption that all vertebrates are evolved from a common ancestral genome and thus should be expected to share much of their genomic structure. Put another way, while the overall structure of a chromosome is expected to change over time due to mutations, many parts of a chromosome will retain their structural integrity over time albeit the more time that passes the more differences are expected.
Take the mammal genome structure as an example. In all genomes examined thus far, the leptin gene is found with the same neighboring genes. Given this strong conservation of genomic architecture it was reasoned that leptin might be found with the same neighbors in other animal genomes. When investigators looked in fish and lizard and found the genes that are next to leptin in mammals, guess what they found next to these genes? Something that looked vaguely like leptin. Once the leptin gene was identified they realized that the DNA sequence of leptin was so radically different in fish (only 13% similar to human or mouse!) that it was understandable why the first attempts to find the gene yielded no result.

Radically different? How then do we know that this gene in fish really is the same gene leptin that is found in mammals? Its position in the genome is the first clue. The gene next to this leptin sequence is the same as the gene found in mammals. But more importantly we can study the protein that this radically different DNA sequence produces. And guess what it makes? A protein that looks and acts just like human/mammalian leptin. That’s right, although the primary DNA sequence is so different and those differences result in a different combination of amino acids, those amino acids fold up into a protein structure that is visually and statistically very similar to mammalian leptin hormone. In fact, human leptin can trigger a response in that fish leptin receptor because it is so similar to fish leptin hormone.

The evolutionary hypothesis to explain these observations is that over 300 million years the fish and mammal leptin sequences have been undergoing mutations to their primary DNA sequences but natural selection has been only accepting mutations that allow for a single functional structure to be formed. There are thousands of combinations of sequences that will generate the same structure and all animals that have a working copy of leptin have one of these combinations. Today we hypothesize that all mammals have nearly the same combination because they have a recent common ancestor and thus they all share a recent common DNA sequence combination for this gene. Remember, nearly all animals make the same functional leptin hormone so sequence similarity is not a requirement for them to produce a functioning hormone.
This brings us back around to birds. If fish and reptiles can have such different DNA sequences but those sequences can essentially make the same leptin hormone, maybe birds simply have another very different DNA sequence combination for making leptin that we haven’t yet recognized. Well, maybe, but there are still more mysteries to probe before I can reveal how we found this elusive gene.
The chicken genome – yet another enigma
The bird leptin could be hiding by virtue of some radical DNA sequence differences but one would not think that it would be able to hide for long given that current technology is allowing us to sequence multiple entire bird genomes in the past year alone. Complete genomes should allow us to reconstruct genome structure and allow the same approach that allowed us to find leptin in fish and reptiles, ie. gene synteny comparisons.
The first bird genome to be completed was the chicken genome. Guess what they didn’t find: Leptin! Leptin researches looked where they would expect to find leptin based on comparisons with gene order in other animals. What they found was a glaring hole in the chicken genome. Not only was leptin missing but the genes that should be next to leptin were also missing in the published chicken genome.
Add to this the fact that researchers have been unable to physically identify the hormone itself in the blood of chickens and it began to appear that chickens, and birds by association, just might not have a leptin gene and thus not make leptin hormone. As recently as 2010, an editorial appeared in the journal General and Comparative Endocrinology entitled “Is there a leptin gene in the chicken genome? Lessons from phylogenetics, bioinformatics and genomics” (Reference 3). The conclusion of the article was that although there were many reasons to expect that chickens should have leptin no physical evidence of the gene and its product had yet been produced. As a result many researchers began to consider that chickens, and maybe all birds, simply have lost the gene for making leptin hormone. I say lost because all other vertebrates have this gene and since birds are thought to be evolved from reptiles their ancestors would have had the gene.
But what about the fact that birds have an apparently functional leptin receptor protein? Doesn’t the existence of a receptor necessitate the existence of the hormone that stimulates the receptor? Not necessarily. There are many examples of genes in both our own genome and other mammals that actively produce a product even though another piece of the metabolic pathway needed to make use of that product doesn’t function correctly. Maybe the chicken leptin receptor is futilely waiting for leptin to stimulate it not knowing the leptin gene has been lost from the genome.
Bird leptin mysteries abound
Leptin is a critical hormone in animals and yet no trace of this hormone has been positively identified in birds. Leptin injected into birds causes an effect but searches for the DNA sequence, traces of the gene product and location within the genome of chickens have provided mixed signals about the existence of leptin in birds.
Now that I have set the stage to this drama, I am ready to tell the story of how the leptin group at the University of Akron found the elusive bird leptin gene. But that story will have to wait until Part II of The Elusive Bird Leptin: And Now for the Rest of the Story.
update: Part II is published and you can find it HERE.
References
1. Prokop, J.W., Schmidt, C, Gasper D, Duff RJ, Ball HC, Ohkubo T, Shawkey MD, Mays HL, Milsted A, Cogburn LA, Londraville RL. 2014 (March). The elusive avian leptin: Identification of several ‘missing links’ in leptin evolution. PLOSOne
2. Londraville, R. L. Y. Macotela, R. J. Duff, M. Easterling, Q. Liu, and E. J. Crespi. 2014 (February). Comparative endrocrinology of leptin: Assessing function in a phylogenetic context. General and Comparative Endrocrinology. Feb 2014. http://dx.doi.org/10.1016/j.ygcen.2014.02.002
3. Pitel F, Faraut T, Bruneau G, Monget P: Is there a leptin gene in the chicken genome? Lessons from phylogenetics, bioinformatics and genomics. Gen Comp Endocrinol 2010;167:1–
4. Prokop, J.W., R.J. Duff, H.C.Ball D.L. Copeland and R.L. Londraville. 2012 (October). Leptin and leptin receptor: Analysis of a structure to function relationship in interaction and evolution from humans to fish. Peptides 38: 326-336.
5. Copeland, D.S., R. Joel Duff, Q. Liu, J. Prokop, and Richard L. Londraville. June 2011. Leptin in teleost fishes: an argument for comparative study. Frontiers in Physiology 2:26:1-11 http://www.frontiersin.org/Journal/Abstract.aspx?s=54&name=aquatic physiology&ART_DOI=10.3389/fphys.2011.00026
Exciting! A nerdy comment: The platypus is a monotreme, which technically should not be included among the marsupials, right? They are an outgroup to all other mammals, if I remember correctly.
LikeLike
Wow, nice catch. Yeah, I have the platypus labeled as a Marsupial. You should have reviewed that manuscript. That’s what happens when you put the botany guy (me) in charge of making a vertebrate phylogeny:-)
LikeLike
What a cliffhanger!
LikeLike
Yeah, I’ll try not to keep you in suspense too long. I’m hoping to get part II done today or tomorrow.
LikeLike
Is a lepton an unusually lean sub-atomic particle? :)
LikeLike
Nice work Joel. Here’s the ref: http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0092751
LikeLike