Few creatures have captured the popular imagination quite like tardigrades. These microscopic eight-legged animals—commonly known as “water bears” or “moss piglets”—are found in virtually every habitat on Earth, from the deep ocean floor to the summit of the Himalayas, from Antarctic ice sheets to the moss growing on the tree in your backyard. What makes them famous, however, is not their ubiquity but their near-indestructibility. Among the 1500 identified species of Tardigrades some can survive being frozen to near absolute zero, heated above the boiling point of water, subjected to the vacuum and radiation of outer space, and blasted with doses of ionizing radiation hundreds of times beyond what would kill a human being. They are, by almost any measure, the toughest animals on the planet.
It should come as no surprise, then, that young-earth creationists have seized upon tardigrades as evidence for fiat divine design. A recent article from Creation Ministries International (CMI) https://creation.com/en/articles/tardigrades-too-tough-for-evolution argues that tardigrades are “too tough for evolution” to have produced by any natural providential process. The core claim is straightforward: natural selection can only favor traits that help an organism survive in environments it actually encounters, so evolution cannot be expected to “over-engineer” a creature for environments it has never faced. Since tardigrades can survive conditions far more extreme than anything found in their natural habitats—the vacuum of space, for instance, or bombardment by X-rays—these abilities must have been directly designed by a Creator rather than produced by natural processes. I can’t help but wonder, is so, for what purpose? Did the Creator intend that water bears should conquer space one day? Or maybe they came from outer space to earth in the past?
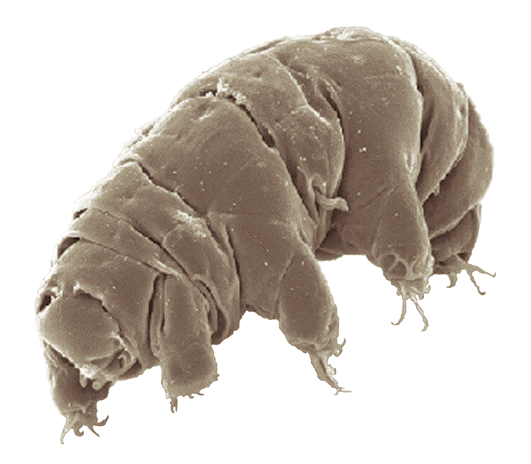
Figure 1 Tardigradum in active state. The body is only 1/100th of an inch long. doi:10.1371/journal.pone.0045682.g00
This design argument for over-engineered features sounds intuitive. It has the feel of common sense. But it rests on a fundamental misunderstanding of how evolution actually works—a misunderstanding so basic that it deserves careful attention, because it is not limited to this one article. The same error recurs throughout creationist literature whenever an organism displays a capacity that seems to exceed the demands of its immediate environment. Let’s take a close look at what is actually going on with tardigrade toughness, and why it poses little challenge to evolutionary theory.
Life Without Water: The Tun State and the Real Selective Pressure
To understand tardigrade resilience, you must first understand their lifestyle. Tardigrades are aquatic animals. Even species that live in terrestrial environments—on mosses, lichens, leaf litter, and soil—require a thin film of water surrounding their bodies to remain active. They feed, move, and reproduce only when hydrated. But the habitats they occupy are frequently subject to drying. A patch of moss on a rock may be drenched by rain one day and baked dry by the sun the next. A lichen crust on a tree trunk can go weeks without moisture.
Many of the 1500 living species of tardigrades display an extraordinary response to this challenge: when their environment dries out, they enter a state of suspended animation called cryptobiosis, or more specifically anhydrobiosis (“life without water”). The animal contracts its body, retracts its legs, and forms a compact barrel-shaped structure called a “tun.” In this state, the tardigrade’s metabolism drops to as little as 0.01% of its normal rate. Special proteins—including the tardigrade-specific cytosolic abundant heat-soluble (CAHS), secretory abundant heat-soluble (SAHS), and mitochondrial abundant heat-soluble (MAHS) proteins—replace much of the water in the animal’s cells, forming a glassy matrix that stabilizes cellular structures and prevents the catastrophic damage that desiccation would normally cause to proteins and membranes.
This is the key insight: the extreme tolerance of tardigrades is not a set of independent adaptations to dozens of separate extreme environments. It is primarily a consequence of adaptations to desiccation—a challenge tardigrades face routinely in their everyday habitats. The same mechanisms that protect cellular structures from the damage of water loss also happen to protect them from many other forms of physical stress.
Consider what happens to a tiny organism when it dries out on a sun-exposed rock. The loss of water concentrates intracellular solutes, generating osmotic stress. The removal of the water layer that normally shields biological molecules exposes them directly to ultraviolet radiation from the sun. Reactive oxygen species accumulate, attacking DNA, proteins, and lipid membranes. The animal may experience temperature extremes as its microhabitat swings from scorching afternoon heat to freezing overnight lows. A tardigrade living on a patch of moss in your garden routinely faces a combination of desiccation, UV exposure, oxidative stress, and temperature fluctuation—all within the span of an ordinary week.
Natural selection has driven tardigrades to evolve robust solutions to this suite of everyday environmental challenges. And here is where the CMI argument falls apart: the molecular mechanisms that protect against desiccation and UV radiation also protect against many forms of extreme stress that tardigrades would never encounter in nature. This is not “over-engineering.” This is an entirely predictable outcome of adaptation to one set of challenges that happens to confer resistance to superficially unrelated challenges, because the underlying molecular damage is similar.
Exaptation: A Frequently Misunderstood Concept
Evolutionary biologists have a well-established term for traits that originally evolved for one function but prove useful in a different context: exaptation. The concept was formally introduced by Stephen Jay Gould and Elisabeth Vrba in 1982, though Darwin himself recognized that the function of a trait can shift over evolutionary time. The classic example is feathers: they originally evolved in dinosaurs for insulation or display, long before any lineage used them for powered flight. When feathers were later co-opted for flight, that was an exaptive use—the trait had not originally been shaped by selection for that purpose.
Tardigrade resilience follows this pattern precisely. The proteins and repair mechanisms that protect tardigrade cells from desiccation and UV radiation happen to also protect them from ionizing radiation, extreme pressure, extreme cold, and the vacuum of space—not because natural selection shaped these mechanisms for those exotic conditions, but because the underlying biochemistry of protection is broadly effective. When a protein stabilizes cellular membranes against the stress of water loss, it also stabilizes them against the stress of extreme cold. When a DNA repair pathway efficiently fixes UV-induced damage, it is also effective against damage from X-rays or gamma rays, because both ultimately generate similar types of molecular lesions—particularly double-strand breaks and hydroxyl radical damage.
The CMI argument assumes that natural selection would need to independently and specifically optimize an organism for every single environment in which it can survive. But that is not how molecular biology works. A robust DNA protection system does not check the source of the incoming damage before deciding whether to function. It simply protects DNA. If the mechanism evolved because UV radiation in a dried tun state selected for it, the protection it provides is not limited to UV radiation alone.
The Dsup Protein: A Case Study in Tardigrade-Unique Innovation
Perhaps the most striking molecular example of tardigrade resilience is the Damage Suppressor protein, or Dsup. First identified in 2016 by Hashimoto and colleagues during their sequencing of the genome of Ramazzottius varieornatus—one of the most stress-tolerant tardigrade species—Dsup is a nuclear protein that physically associates with chromatin, the packed form of DNA in the cell nucleus. When researchers engineered human cells to express Dsup, those cells showed approximately 40% less DNA fragmentation after X-ray exposure compared to normal cells. The Dsup-expressing cells even retained their ability to proliferate after irradiation doses that would normally destroy a cell’s reproductive capacity.
Subsequent research has revealed that Dsup works by binding to nucleosomes—the protein spools around which DNA is wound—and forming a kind of molecular shield against hydroxyl radicals, the chemically reactive fragments produced when radiation interacts with water inside cells. A 2019 study published in eLife demonstrated that Dsup from R. varieornatus contains a region with structural similarity to the nucleosome-binding domain of vertebrate HMGN proteins, though the two protein families have quite low overall sequence similarity. Intriguingly, a Dsup ortholog was subsequently found in another tardigrade species, Hypsibius exemplaris, with only about 26% amino acid identity to the R. varieornatus version—yet it functions similarly, binding nucleosomes and protecting DNA from hydroxyl radical damage.
The Dsup protein is intrinsically disordered—it does not fold into a rigid three-dimensional structure but instead exists as a flexible, highly charged mass of protein that drapes across the chromatin and absorbs chemical insults. It is enriched in serine, alanine, glycine, and lysine residues (collectively called “SAGK” residues), which are amino acids that promote this disordered state. Molecular dynamics simulations suggest that Dsup’s inherent flexibility allows it to adjust its shape to fit the contours of DNA, providing both structural support and a kind of electrical shielding against reactive oxygen species.
Now, here is what the CMI article conspicuously fails to mention: Dsup’s protective function against ionizing radiation is almost certainly a secondary benefit of a protein that evolved to protect DNA during the oxidative stress of desiccation and UV exposure. When a tardigrade enters the tun state and loses its protective water layer, it is directly exposed to environmental UV radiation, which generates precisely the kind of hydroxyl radical damage that Dsup is designed to suppress. A tardigrade sitting as a dried tun on a sun-baked rock faces intense UV bombardment—a challenge that has recurred billions of times over hundreds of millions of years of tardigrade evolution. This is the selective pressure. The ability to also survive X-rays in a laboratory is a byproduct—an exaptation—of a protein shaped by selection for a very real and very ordinary environmental challenge.
Research published in BMC Genomics in 2022 by Yoshida and colleagues provides direct support for this interpretation. Their time-series transcriptomic analysis of R. varieornatus revealed extensive overlap between the genes activated in response to UV radiation and those activated during anhydrobiosis. The same anti-oxidative stress pathways, the same DNA repair genes, the same tardigrade-specific protection proteins—including CAHS and SAHS family members—are upregulated under both conditions. The molecular machinery of desiccation tolerance and UV tolerance is not merely similar; in many cases it is identical. Desiccation tolerance and radiation tolerance are two sides of the same evolutionary coin.
Tardigrades Are Not the Only Extremotolerant Organisms—and That Matters
The CMI article presents tardigrade resilience as a singular marvel inexplicable by natural processes. But tardigrades are far from the only organisms with extraordinary stress tolerance. Deinococcus radiodurans, a bacterium, can survive radiation doses thousands of times greater than the human lethal dose—and its radiation tolerance is understood to have evolved as a byproduct of its remarkable desiccation tolerance. Bdelloid rotifers, another group of microscopic aquatic animals, can survive extreme desiccation, radiation, and even the vacuum of space, using mechanisms that include both robust DNA repair systems and—in a fascinating parallel to tardigrades—the acquisition of foreign genes through horizontal gene transfer. The resurrection plant Selaginella lepidophylla can lose virtually all its water content and revive when rehydrated, surviving temperature and radiation extremes along the way. Brine shrimp eggs (cysts of Artemia) show similar cross-tolerance between desiccation and radiation.
In every one of these cases, the pattern is the same: organisms that evolved robust desiccation tolerance also display tolerance to radiation, temperature extremes, and other stresses they rarely or never encounter in their natural habitats. This is not a parade of independent design miracles. It is a consistent and predictable consequence of the biochemistry of stress protection. The molecular damage caused by desiccation, UV radiation, ionizing radiation, and extreme temperatures all converge on the same cellular targets: DNA strand breaks, protein denaturation, lipid membrane disruption, and oxidative damage from reactive oxygen species. An organism that evolves powerful defenses against any one of these stresses will inevitably display cross-tolerance to others, because the underlying damage pathways overlap.
The creationist argument requires you to ignore this convergent pattern across unrelated lineages and instead view each case as a separate act of special creation. It requires you to ask, “Why would evolution give a tardigrade the ability to survive in space?” while ignoring the much more productive question: “What everyday environmental challenges have tardigrades faced over their evolutionary history that would select for the molecular machinery we observe?” The answer to that second question is well established: repeated cycles of desiccation and rehydration in UV-exposed terrestrial and semi-terrestrial habitats, stretching back at least 180 million years and possibly more than 400 million years, based on fossil calibrations of the tardigrade molecular clock published in Communications Biology in 2024.
The Evolutionary History of Tardigrade Extremotolerance
The CMI article also obscures the rich evolutionary context of tardigrade resilience. Far from being a static suite of abilities that appeared all at once, tardigrade stress tolerance has a complex evolutionary history that researchers are actively reconstructing.
Tardigrades are a diverse group of species that belong to the Ecdysozoa, the superphylum of molting animals that also includes arthropods and nematodes. They are descended from lobopodians—an informal group of ancient marine worms with stubby legs—that lived during the Cambrian Period, roughly 500 million years ago. Modern tardigrades appear to have transitioned from marine to terrestrial habitats at least twice independently: once in the lineage leading to the eutardigrades and once in the heterotardigrades. This transition any some tardigrades to land—and the attendant challenge of desiccation—is widely understood to be the primary selective driver behind the evolution of anhydrobiosis.
A 2024 study by Fleming, Pisani, and Arakawa published in Genome Biology and Evolution produced the first phylogenetic analyses of six tardigrade protein families linked to desiccation and radiation tolerance. Their results revealed numerous independent gene duplications across different tardigrade lineages, indicating that extremotolerance did not arise in a single event but was assembled piecemeal through multiple independent adaptations as different lineages colonized increasingly arid habitats. The tardigrade-specific CAHS, SAHS, and MAHS protein families, for example, show complex patterns of lineage-specific expansion that correlate with habitat transitions.
Even the Dsup protein has a history that speaks to gradual evolution rather than sudden appearance. Dsup is found in R. varieornatus and a Dsup-like ortholog exists in the related species H. exemplaris, but syntenic analysis of the genomic regions surrounding these genes shows conserved flanking genes, supporting the interpretation that Dsup arose within the tardigrade lineage and has been evolving since the divergence of these species. The low sequence identity (about 26%) between the two versions, combined with conservation of the functionally critical HMGN-like nucleosome-binding domain, is a classic signature of a protein under selection for function while diverging in overall sequence—exactly what we expect from ordinary molecular evolution.
Notably, Dsup is absent from some tardigrade species, and its presence correlates with particularly high radiation tolerance. H. exemplaris (previously classified as H. dujardini) shows lower desiccation tolerance and lower UV tolerance than R. varieornatus, though it still displays remarkably high tolerance to gamma radiation in the hydrated state. This variation among species is precisely what we would expect if extremotolerance is the product of ongoing, lineage-specific evolutionary refinement rather than a one-time design decision. If tardigrade resilience were the product of direct divine engineering, why would different species vary so dramatically in their tolerance profiles? Why would some species lack Dsup entirely?
The Real Misunderstanding: Confusing Current Utility with Historical Origin
At its root, the CMI argument commits a fundamental error that has been recognized and discussed in evolutionary biology for well over a century: it confuses the current utility of a trait with the historical selective pressures that shaped it. Darwin himself noted that the function of a trait can shift over its evolutionary history. Gould and Vrba gave this phenomenon a name. And the tardigrade literature is virtually unanimous in recognizing that radiation tolerance, vacuum tolerance, and pressure tolerance are byproducts of adaptations to desiccation and the oxidative stress that accompanies it.
The CMI article frames this as a devastating problem for evolution: “Natural selection can only select characteristics necessary for immediate survival. Consequently, evolution cannot be expected to over-engineer creatures for a host of environments they have never faced.” But this framing presupposes that each extreme tolerance must have been independently selected for, which is precisely the assumption that the evidence contradicts. The tardigrade has not been “over-engineered.” It has been precisely engineered—by natural selection—for survival in habitats where desiccation, UV exposure, and oxidative stress are constant threats. The ability to also survive in space is not a separate feature requiring its own explanation; it is an automatic consequence of the features that do have direct selective explanations.
The CMI article also claims that “proteins around DNA would normally hinder reproduction; so, while the (designed) tardigrade Dsup brilliantly avoids this, selection would work against a partly-evolved version.” This is presented as an argument against the gradual evolution of Dsup, but it is undermined by the actual research. Dsup is an intrinsically disordered protein—it does not form a rigid barrier around DNA but rather a flexible, dynamic cloud that can be displaced during replication and transcription. Indeed, studies expressing Dsup in Drosophila melanogaster cells (published in iScience in 2023) found that while Dsup does interact with DNA in a non-specific way that can influence transcription, the protein still confers measurable radioprotective benefits. The relationship between DNA protection and transcriptional interference is a quantitative tradeoff, not an all-or-nothing binary—precisely the kind of tradeoff that natural selection navigates routinely.
What Would the Young-Earth Model Actually Predict?
It is worth pausing to consider what we would expect to observe if tardigrade resilience were the product of direct divine creation, as the CMI article implies. Presumably, a designed system would be optimally tuned for the specific challenges the organism would face in its intended habitat. We might expect uniform resilience across all tardigrade species—after all, if the abilities were deliberately installed rather than gradually evolved, there is no obvious reason for such dramatic variation among species. We would not necessarily expect the molecular machinery of radiation tolerance to be identical to the machinery of desiccation tolerance; a designer might use distinct mechanisms for distinct challenges.
Instead, what we observe is precisely what evolutionary theory predicts: a complex patchwork of tolerance abilities that varies across species in ways that correlate with habitat and lifestyle. Species that face more frequent desiccation tend to be more tolerant of radiation. The same genes and proteins are deployed against both desiccation and radiation stress. Tolerance mechanisms show clear phylogenetic signatures—they cluster by evolutionary relatedness, not by habitat type alone. Gene families have undergone lineage-specific duplications and losses that map onto habitat transitions. And the entire system is built from the same basic molecular toolkit—antioxidant defenses, DNA repair, intrinsically disordered proteins—that is used by other desiccation-tolerant organisms across the tree of life, from bacteria to plants to rotifers.
None of this looks like special immediate creation. All of it looks like the result of a process of adaptation to environmental challenges.
A Better Way to Read Both Scripture and Nature
As a Christian and a biologist, I find the tardigrade story to a beautiful illustration of how God’s creation works through secondary causes—through the ordinary, sustained processes of natural selection operating over deep time—to produce results that inspire genuine wonder. The tardigrade does not need to have been individually engineered for every extreme condition it can survive in order to be a testament to the wisdom embedded in creation. The fact that a robust system of desiccation protection automatically confers resistance to radiation, vacuum, and extreme temperatures is itself a reflection of the deep coherence of the physical and chemical laws that govern the natural world—laws that my Christian tradition has always understood as the faithful, ongoing expression of God’s providential governance.
What troubles me about the CMI argument is not merely that it gets the science wrong—though it does, quite badly. What troubles me more is the implicit theology: the assumption that God’s creative work must take the form of direct, unmediated intervention at every point, and that the operation of natural processes is somehow a rival to divine agency rather than its instrument. This is not the theology of the Reformed confessions. It is not the theology of Calvin, who wrote that God “daily discloses himself in the whole workmanship of the universe” through natural means. And it is not a theology that will serve believers well when they encounter the actual evidence for how tardigrades—and the rest of the living world—came to be as they are.
The tardigrade is not too tough for evolution. It is exactly as tough as its evolutionary history would lead us to expect—and the details of how it achieved that toughness are far more interesting and far more theologically suggestive than the simplistic design argument allows.
Blessings, Joel
References
Beltran-Pardo, E., Jönsson, K.I., Harms-Ringdahl, M., Haghdoost, S., & Wojcik, A. (2015). Tolerance to gamma radiation in the tardigrade Hypsibius dujardini from embryo to adult correlate inversely with cellular proliferation. PLOS ONE, 10(7), e0133658.
Chavez, C., Cruz-Becerra, G., Fei, J., Bhanu, N.V., et al. (2019). The tardigrade damage suppressor protein binds to nucleosomes and protects DNA from hydroxyl radicals. eLife, 8, e47682.
Fleming, J.F., Pisani, D., & Arakawa, K. (2024). The evolution of temperature and desiccation-related protein families in Tardigrada reveals a complex acquisition of extremotolerance. Genome Biology and Evolution, 16(1), evad217.
Gould, S.J. & Vrba, E.S. (1982). Exaptation—a missing term in the science of form. Paleobiology, 8(1), 4–15.
Hashimoto, T., Horikawa, D.D., Saito, Y., et al. (2016). Extremotolerant tardigrade genome and improved radiotolerance of human cultured cells by tardigrade-unique protein. Nature Communications, 7, 12808.
Hashimoto, T. & Kunieda, T. (2017). DNA protection protein, a novel mechanism of radiation tolerance: Lessons from tardigrades. Life, 7(2), 26.
Jönsson, K.I. (2019). Radiation tolerance in tardigrades: Current knowledge and potential applications in medicine. Cancers, 11(9), 1333.
Kamilari, M., Jørgensen, A., Schiøtt, M., & Møbjerg, N. (2019). Comparative genome analysis of the tardigrades Hypsibius dujardini and Ramazzottius varieornatus. PLOS Biology, 15(7), e2002266.
Mapalo, M.A., et al. (2024). Revised tardigrade family tree and new insights into the evolution of cryptobiosis. Communications Biology.
Merziak, A., et al. (2023). The tardigrade Dsup protein enhances radioresistance in Drosophila melanogaster and acts as an unspecific repressor of transcription. iScience, 26(7), 106998.
Yoshida, Y., Satoh, T., Ota, C., et al. (2022). Time-series transcriptomic screening of factors contributing to the cross-tolerance to UV radiation and anhydrobiosis in tardigrades. BMC Genomics, 23, 405.